Introduction
Protein domains are highly useful to better understand the construction and function of a protein [1]. Each protein domain will have its own function and association with biological process. Oftentimes proteins will contain multiple domains, and segments can even be repeated and interact with each other [2]. Protein domains can also have categorized Gene Ontology (GO) Terms to help examine its functions.
results
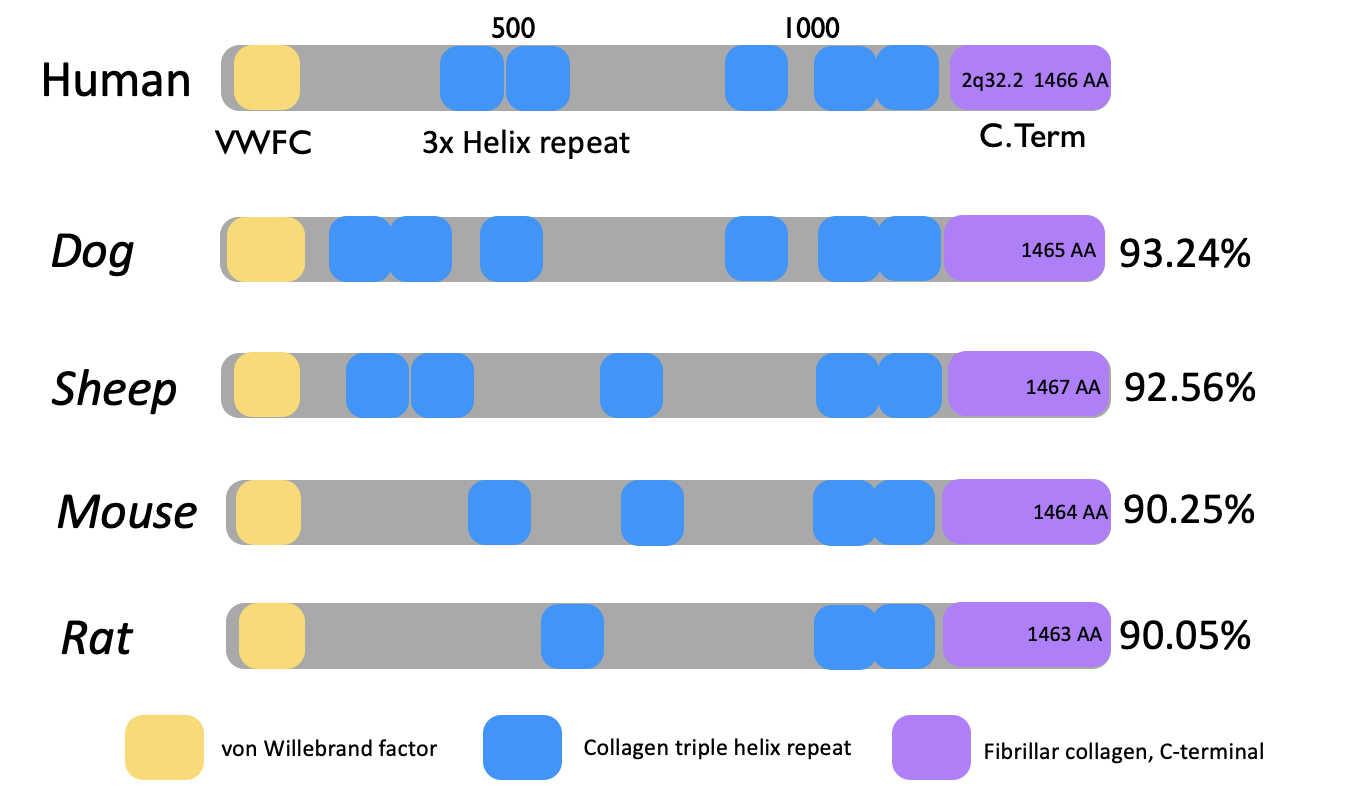
Protein domains of the COL3A1 protein
Conclusion
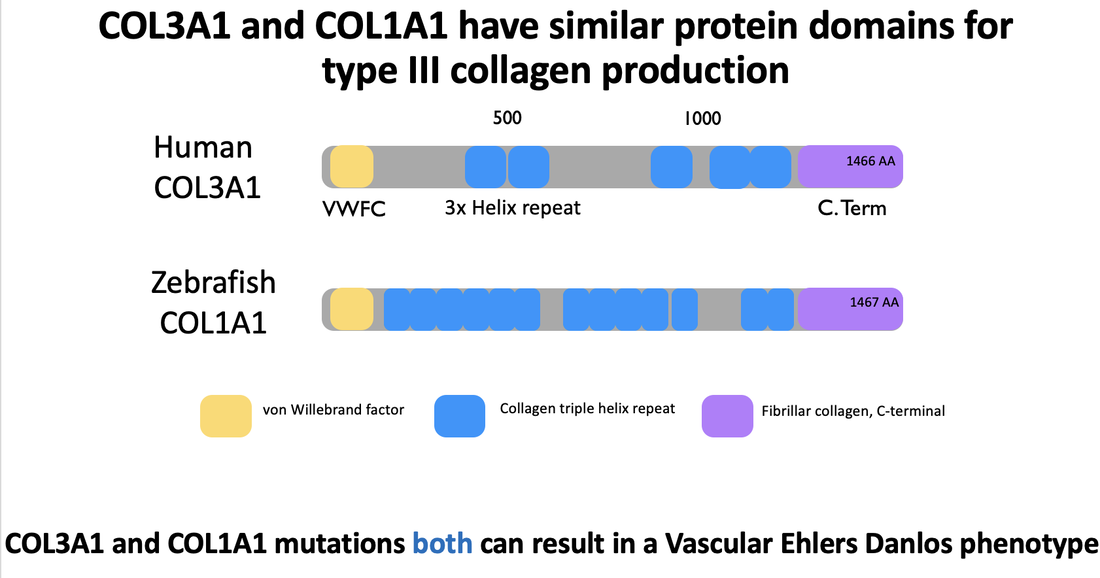
There is a high percent identity seen throughout model organisms for COL3A1. Furthermore, the COL3A1 protein domains in humans are highly similar to COL1A1 in Zebrafish. Zebrafish do not express the COL3A1 protein, but the COL1A1 proteins shares a similar domain structure and homology to that of COL3A1 in humans.
Here is more information about the protein domain factors seen in COL3A1 and COL1A1:
The von Willebrand Factor: Proteins containing this domain are often extracellular involved with biological processes such as cellular adhesion and migration [3]. Many fibril collagen proteins (including COL3A1 and COL1A1) express this factor.
Collagen triple helix repeat: This domain is critical for the formation of fibril collagens [4]. It contains the (G-X-Y) repeats and polypeptides to form the characterized triple helix. X and Y have potential to be any reside, but proline and hydroxyproline are common.
Fibrillar collagen, C-terminal: There is a high conservation seen in this domain for fibril collagen proteins [5]. It is useful for the assembly of the collagen fibrils themselves.
Here is more information about the protein domain factors seen in COL3A1 and COL1A1:
The von Willebrand Factor: Proteins containing this domain are often extracellular involved with biological processes such as cellular adhesion and migration [3]. Many fibril collagen proteins (including COL3A1 and COL1A1) express this factor.
Collagen triple helix repeat: This domain is critical for the formation of fibril collagens [4]. It contains the (G-X-Y) repeats and polypeptides to form the characterized triple helix. X and Y have potential to be any reside, but proline and hydroxyproline are common.
Fibrillar collagen, C-terminal: There is a high conservation seen in this domain for fibril collagen proteins [5]. It is useful for the assembly of the collagen fibrils themselves.
References
[1] EMBL-EBI. (2023). What are protein domains? | Protein classification. EMBL-EBI. https://www.ebi.ac.uk/training/online/courses/protein-classification-intro-ebi-resources/protein-classification/what-are-protein-domains/
[2] Wang Y, Zhang H, Zhong H, Xue Z. Protein domain identification methods and online resources. Comput Struct Biotechnol J. 2021;19:1145-1153. Published 2021 Feb 2. doi:10.1016/j.csbj.2021.01.041
[3] VWFC domain InterPro Entry. (n.d.). Www.ebi.ac.uk; EMBL’s European Bioinformatics Institute. Retrieved April 09, 2024, from https://www.ebi.ac.uk/interpro/entry/InterPro/IPR001007/
[4] Collagen triple helix repeat InterPro Entry. (n.d.). Www.ebi.ac.uk; EMBL’s European Bioinformatics Institute. Retrieved April 09, 2024, from https://www.ebi.ac.uk/interpro/entry/InterPro/IPR001007/
[5] Fibrillar collagen, C-terminal InterPro Entry. (n.d.). Www.ebi.ac.uk; EMBL’s European Bioinformatics Institute. Retrieved April 10, 2024, from https://www.ebi.ac.uk/interpro/entry/InterPro/IPR001007/
[2] Wang Y, Zhang H, Zhong H, Xue Z. Protein domain identification methods and online resources. Comput Struct Biotechnol J. 2021;19:1145-1153. Published 2021 Feb 2. doi:10.1016/j.csbj.2021.01.041
[3] VWFC domain InterPro Entry. (n.d.). Www.ebi.ac.uk; EMBL’s European Bioinformatics Institute. Retrieved April 09, 2024, from https://www.ebi.ac.uk/interpro/entry/InterPro/IPR001007/
[4] Collagen triple helix repeat InterPro Entry. (n.d.). Www.ebi.ac.uk; EMBL’s European Bioinformatics Institute. Retrieved April 09, 2024, from https://www.ebi.ac.uk/interpro/entry/InterPro/IPR001007/
[5] Fibrillar collagen, C-terminal InterPro Entry. (n.d.). Www.ebi.ac.uk; EMBL’s European Bioinformatics Institute. Retrieved April 10, 2024, from https://www.ebi.ac.uk/interpro/entry/InterPro/IPR001007/
This web page was produced as an assignment for Genetics 564, a capstone course at UW-Madison